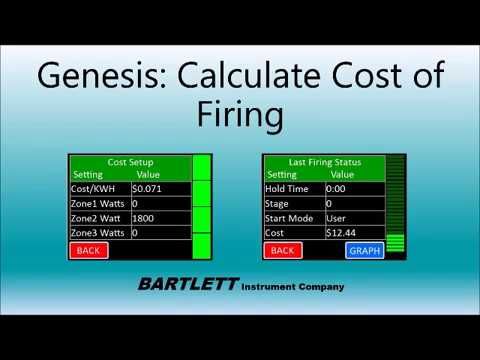
Choose Your Kiln Model
Shop Our Kilns

Filter by Category
Paragon News
| Image | Name | Weight | Outer Dimensions | Price | Buy | hf:tax:function_facet | hf:tax:feature_facet | hf:tax:size_facet | hf:tax:shape_facet |
|---|
-
BlueBird Glass Bead Annealing Kiln
$1,548.68 -
BlueBird XL Glass Annealing Paragon Kiln
$1,883.52 -
Caldera Multi Purpose Digital Electric Kiln
$1,340.44 -
Caldera XL Multi Purpose Digital Electric Kiln
$1,553.71 -
CS14D Dual Clam Shell Glass Fusing Kiln
$2,243.66 -
CS14S Clam Shell Glass Fusing Kiln
$2,066.46 -
Dragon 24 Front Loading Kiln with Stand
$11,130.12 -
E10 Enameling and China Painting Medium Kiln
$1,660.24 -
E12 Enameling and China Painting Medium Kiln
$1,673.16 -
E12T Enameling and China Painting Large Kiln
$1,853.23 -
E14 Enameling and China Painting XL Kiln
$2,273.39 -
E9 Enameling and China Painting Small Kiln
$1,403.46 -
F Series
Starting at $2,412.44 -
F130 Glass Bead Annealing and Lamp Working Kiln
$2,769.43 -
FiberFuse 16 Glass Fusing Kiln
$2,268.96 -
Home Artist Portable Raku Kiln
$2,028.52 -
HT Series
Starting at $2,529.94 -
Iguana Front Loading Kiln with Stand
$6,110.91 -
KM14 Pro Knife Making Paragon Kiln
$4,302.53 -
KM18 Pro 3-Zone Knifemaking Kiln
$4,583.64 -
KM24 Pro 3-Zone Knifemaking Kiln
$5,295.97 -
KM24 Pro Series
Starting at $5,295.97 -
KM24 Series
Starting at $2,284.20 -
KM36 Pro 3-Zone Knifemaking Kiln
$6,499.41 -
KM36T Pro Series
Starting at $6,499.41 -
KM36T Series
Starting at $2,549.78 -
KM45 Pro 3-Zone Knifemaking Kiln
$7,870.93 -
KM45 Series
Starting at $4,229.31 -
PKM9D Double Barrel Heat Treating Kiln
$2,664.96 -
SC2 FIBER JEWELRY DIGITAL ELECTRIC KILN
$1,387.97 -
SC2 PRO FIBER JEWELRY DIGITAL ELECTRIC KILN
$1,591.85 -
SC3 Fiber Jewelry Digital Electric Kiln
$1,579.12 -
Super Dragon Kiln with Stand
$18,553.19 -
TNF1613 Series
Starting at $2,070.20 -
TNF233 10 Sided Ceramic and Pottery Kiln
$3,914.25 -
TNF243 10 Sided Ceramic and Pottery Kiln
$4,566.62 -
TNF273 12 Sided Ceramic & Pottery Kiln
$4,825.84 -
TNF283 Series
Starting at $4,996.88 -
TNF823 8 Sided Ceramic and Pottery Kiln
$3,010.58 -
Viking28 Series
Starting at $5,866.21 -
Xpress 66 7 Sided Ceramic and Pottery
$1,794.34